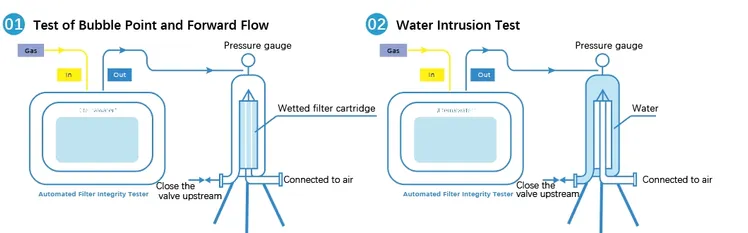
How to Process Bubble Point Testing for Hydrophobic Filters?

1. Why Bubble Point Testing Is Essential for Hydrophobic Filters
Hydrophobic filters are widely used in critical filtration applications where gas flow, sterile venting, or moisture exclusion is required. Typical applications include sterile air filtration in pharmaceutical manufacturing, tank venting systems, compressed air purification, and gas filtration in food and beverage processing. In these applications, filter integrity is not optional—it is a fundamental requirement to ensure process safety and product quality.
Bubble point testing is one of the most commonly used integrity testing methods for membrane-based filters. It provides a direct, non-destructive way to verify whether the membrane structure remains intact and whether the effective pore size distribution meets design specifications. For hydrophobic filters, bubble point testing plays an even more critical role due to the inherent difficulty of wetting hydrophobic membranes and the higher risk of undetected defects.
A damaged hydrophobic filter may allow unfiltered gas to bypass the membrane, potentially introducing contaminants, microorganisms, or particles into downstream systems. Unlike liquid filtration, gas filtration failures are often invisible and may remain undetected until contamination has already occurred. Bubble point testing helps mitigate this risk by providing measurable, repeatable data that confirms membrane integrity before use.
In regulated industries such as pharmaceuticals and biotechnology, bubble point testing is often integrated into quality control protocols and validation processes. Although alternative integrity tests exist, bubble point testing remains one of the most intuitive and widely accepted methods, particularly for membrane cartridge filters used in sterile gas applications.
2. Differences Between Hydrophobic and Hydrophilic Filters in Bubble Point Testing
Understanding the differences between hydrophobic and hydrophilic filters is essential before discussing how to process bubble point testing correctly.
Hydrophilic filters naturally attract water and can be easily wetted using deionized water or aqueous solutions. In contrast, hydrophobic filters repel water due to their low surface energy and high contact angle. Common hydrophobic membrane materials include polytetrafluoroethylene (PTFE) and certain grades of polyvinylidene fluoride (PVDF).
This fundamental difference significantly affects bubble point testing procedures.
For hydrophilic filters, the membrane pores are readily filled with liquid, creating a stable liquid barrier within the pore structure. When gas pressure is gradually applied, the bubble point corresponds to the pressure required to displace the liquid from the largest pore, forming a continuous stream of bubbles.
Hydrophobic filters, however, cannot be reliably wetted with water alone. If water is used as the wetting liquid, incomplete wetting will occur, leading to artificially low bubble point values or inconsistent results. Therefore, hydrophobic filters require specific wetting liquids—typically low-surface-tension alcohols or alcohol-water mixtures—to ensure complete pore wetting prior to testing.
Another key difference lies in test sensitivity. Hydrophobic membranes often exhibit higher bubble point pressures compared to hydrophilic membranes of similar nominal pore size due to differences in surface tension and wetting behavior. As a result, test parameters such as pressure ramp rate, stabilization time, and bubble detection method must be carefully controlled to obtain accurate and repeatable results.
Recognizing these differences is critical when processing bubble point testing for hydrophobic filters, as applying hydrophilic testing assumptions can easily lead to misinterpretation of results.

3. Physical Principles Behind Bubble Point Testing
Bubble point testing is based on well-established physical principles involving surface tension, capillary action, and pressure equilibrium within porous structures.
At the core of the bubble point test is the relationship between pressure, surface tension, and pore diameter, commonly described by the Laplace equation:
ΔP=4γcosθD\Delta P = \frac{4 \gamma \cos \theta}{D}ΔP=D4γcosθ
Where:
ΔP is the pressure difference required to displace the liquid,
γ is the surface tension of the wetting liquid,
θ is the contact angle between the liquid and membrane material,
D is the effective pore diameter.
For hydrophobic membranes, the contact angle (θ) between the wetting liquid and the membrane surface is a critical factor. Hydrophobic materials exhibit large contact angles with water, which is why water alone is unsuitable for testing. By selecting an appropriate wetting liquid with lower surface tension and improved wetting characteristics, the contact angle can be reduced, allowing the liquid to fully occupy the membrane pores.
During the test, gas pressure is applied gradually to the upstream side of the wetted filter. As pressure increases, gas begins to displace liquid from the largest pore first. The bubble point is defined as the pressure at which a continuous stream of bubbles emerges from the downstream side, indicating that the largest pore has been emptied of liquid.
It is important to distinguish between isolated bubbles and a continuous bubble stream. Isolated bubbles may appear at lower pressures due to surface imperfections or incomplete wetting, but they do not represent the true bubble point. Proper test execution focuses on identifying the sustained bubble flow that corresponds to the largest effective pore.
Understanding these physical principles is essential for interpreting bubble point test results accurately and for diagnosing abnormal readings during hydrophobic filter testing.
4. Process Flow of Bubble Point Testing for Hydrophobic Filters
Processing bubble point testing for hydrophobic filters involves a structured sequence of steps designed to ensure complete wetting, controlled pressurization, and reliable bubble detection. Although specific procedures may vary depending on industry standards and equipment, the general process flow remains consistent.
4.1 Wetting Phase
The wetting phase is the most critical step in bubble point testing for hydrophobic filters. An appropriate wetting liquid must be selected to fully penetrate the membrane pores. Common wetting liquids include isopropyl alcohol (IPA), ethanol, or defined alcohol-water mixtures.
The filter cartridge is immersed, flushed, or circulated with the wetting liquid to remove trapped air and ensure complete saturation. Adequate wetting time is essential, as insufficient wetting can lead to false low bubble point readings.
4.2 Pressurization Phase
After wetting, the filter is connected to the bubble point test apparatus. Gas pressure—typically air or nitrogen—is applied gradually to the upstream side. The pressure ramp rate should be controlled and consistent to avoid overshooting the true bubble point.
During this phase, operators closely observe the downstream side of the filter, which may be submerged in liquid or connected to a bubble detection device.
4.3 Bubble Detection Phase
As pressure increases, individual bubbles may begin to appear. These isolated bubbles should not be mistaken for the bubble point. The true bubble point is reached only when a continuous, steady stream of bubbles is observed.
The pressure at this point is recorded as the bubble point pressure. This value is then compared with manufacturer specifications or established acceptance criteria to determine whether the filter passes the integrity test.
Learn More: Step-by-Step Procedure for Bubble Point Filter Integrity Test
5. Key Parameters Affecting Bubble Point Test Results
Accurate bubble point testing for hydrophobic filters depends on controlling several critical parameters. Even when the correct procedure is followed, variations in these parameters can significantly influence test results and lead to incorrect conclusions regarding filter integrity.
5.1 Wetting Liquid Properties
The surface tension of the wetting liquid directly affects bubble point pressure. For hydrophobic filters, low-surface-tension liquids such as isopropyl alcohol or ethanol are commonly used. Alcohol-water mixtures may also be employed to balance wetting efficiency and safety considerations.
Inconsistent wetting liquid composition can result in variations in surface tension, leading to inconsistent bubble point values. For this reason, standardized wetting fluids with controlled concentrations are recommended, especially in regulated environments.
5.2 Temperature Effects
Temperature influences both the surface tension of the wetting liquid and the viscosity of the gas. As temperature increases, surface tension decreases, which may reduce the measured bubble point pressure. To ensure repeatability, bubble point testing should be conducted at a controlled and documented temperature.
In critical applications, manufacturers often specify acceptable temperature ranges for integrity testing to minimize variability.
5.3 Pressure Ramp Rate
The rate at which pressure is increased during testing plays a crucial role in identifying the true bubble point. A rapid pressure increase may cause the operator to overshoot the actual bubble point, while an excessively slow ramp may lead to evaporation of the wetting liquid or unstable readings.
A controlled, linear pressure increase allows sufficient time for observation and ensures that the recorded bubble point corresponds to the largest effective pore.
5.4 Membrane Material and Structure
Different hydrophobic membrane materials exhibit different wetting behaviors and pore geometries. PTFE membranes, for example, typically require more aggressive wetting conditions than PVDF membranes due to their higher hydrophobicity.
Additionally, membrane thickness, pore shape, and pleat structure can influence gas flow dynamics during testing. These factors must be considered when comparing results across different filter types or manufacturers.
6. Data Analysis and Result Validation
Once the bubble point pressure has been recorded, proper analysis and validation of the data are essential to determine filter integrity accurately.
6.1 Typical Bubble Point Values
Manufacturers usually provide nominal bubble point values or minimum acceptance criteria for each filter grade. These values are derived from controlled laboratory testing and correlated with membrane pore size distribution.
For hydrophobic filters, acceptance criteria are often defined as a minimum bubble point pressure rather than an exact value, allowing for normal test variability while still ensuring membrane integrity.
6.2 Identifying Abnormal Results
Abnormally low bubble point values may indicate:
Incomplete wetting of the membrane
Membrane damage or defects
Improper test setup or leaks
Conversely, unusually high bubble point values may result from:
Incorrect wetting liquid selection
Blocked pores due to contamination
Measurement errors
When abnormal results occur, retesting after verifying wetting conditions and equipment setup is recommended before concluding that the filter has failed.
6.3 Correlation with Other Integrity Tests
In some applications, bubble point testing is complemented by diffusion or pressure hold tests. Correlating results from multiple integrity test methods can provide additional confidence in filter performance, particularly for high-risk or regulated processes.

7. Practical Applications in Industrial Filtration Systems
Bubble point testing for hydrophobic filters is widely applied across multiple industries where gas filtration and sterile venting are critical.
7.1 Pharmaceutical and Biotechnology Applications
In pharmaceutical manufacturing, hydrophobic filters are commonly used for sterile air and gas filtration, fermenter venting, and tank protection. Bubble point testing is often incorporated into validation protocols to ensure compliance with GMP requirements.
7.2 Food and Beverage Processing
Hydrophobic vent filters protect storage tanks and processing vessels from airborne contamination while allowing pressure equalization. Bubble point testing ensures that these filters maintain their integrity throughout repeated cleaning and sterilization cycles.
7.3 Industrial Gas and Compressed Air Systems
In industrial environments, hydrophobic filters are used to remove moisture and particulates from compressed air and process gases. Regular bubble point testing helps prevent system contamination and equipment damage caused by filter failure.
These practical applications demonstrate the versatility and importance of bubble point testing as a routine integrity verification tool for hydrophobic filters.
Conclusion
Bubble point testing is a critical integrity testing method for hydrophobic filters used in gas filtration, sterile venting, and moisture-sensitive applications. Due to the unique wetting challenges associated with hydrophobic membranes, proper test execution requires careful attention to wetting liquids, test parameters, and result interpretation.
By understanding the physical principles behind bubble point testing and following a structured testing process, operators can reliably verify membrane integrity and ensure consistent filtration performance. When integrated into quality control and validation programs, bubble point testing provides a robust and non-destructive means of safeguarding critical filtration systems.
For manufacturers, end users, and quality professionals alike, mastering how to process bubble point testing for hydrophobic filters is essential to maintaining product quality, regulatory compliance, and operational reliability.
References
ASTM F316 – Standard Test Methods for Pore Size Characteristics of Membrane Filters by Bubble Point and Mean Flow Pore Test
- The Importance of Bubble Point Test for Filter Integrity – Econe Filtration
ISO 2942 – Hydraulic Fluid Power — Filter Elements — Verification of Fabrication Integrity and Determination of the First Bubble Point
PDA Technical Report No. 26 – Sterilizing Filtration of Liquids
ASTM F838 – Standard Test Methods for Determining Bacterial Retention of Membrane Filters
Jornitz, M. W., & Meltzer, T. H., Filtration and Separation Technology, Elsevier
Uhlmann, D. R., Surface Tension and Wetting in Porous Materials, Journal of Membrane Science